The CBSE Class 11 Chemistry Lab Manual is provided to the students so they can score well on the examinations. For a subject like Chemistry, it is essential to remember the right reactions and what they result in is vital. The students must try to get full marks in this practical section to increase their overall marks in the examination. Students should study the laws and theories before performing the experiments and are suggested to revise their helpful notes before their examination. That’s why we are providing a Class 11 Chemistry Lab Manual for practice purposes to obtain a great score in the final examination.
[Index]
Overview
Before we discuss the Class 11 Lab Manual, let us check the CBSE Class 11 Summary. Below, we have mentioned the complete CBSE Class 11 Summary. The student is advised to check out to complete the summary.
| Particular | Description |
| Class | 11th |
| Board | CBSE |
| Category | Lab Manual |
| Study Materials | Click Here |
| Class 11 Free E-Book | Click Here |
| YouTube Channel | Click Here |
Class 11 Chemistry Lab Manual
Belo, we have mentioned the CBSE Class 11 Chemistry Lab Manual. Students have checked the complete Class 11 Chemistry Lab Manual in PDF for a great score in the final examination.
Example for Lab Manual

NOTE: The links are given below for downloading the Class 11 Chemistry Lab Manual in PDF format.
| Unit Number | Unit Name |
| Unit 1 | Introduction |
| Unit 2 | Basic Laboratory Techniques |
| Unit 3 | Purification And Criteria Of Purity |
| Unit 4 | Chemical Equilibrium (Ionic Equilibrium In Solutions) |
| Unit 5 | pH and pH change in Aqueous Solutions |
| Unit 6 | Titrimetric Analysis |
| Unit 7 | Systematic Qualitative Analysis |
Class 12 Chemistry Lab Manual Projects
| S.No. | Project No. |
| 1 | Projects 1 to 8 |
Class 11 Chemistry Syllabus 2024-25
Check out the latest CBSE NCERT Class 11 Chemistry Syllabus. The syllabus is for the academic year 2024-25 sessions. First of all, check the CBSE Class 11 Chemistry Exam Pattern. Students should check the complete syllabus and exam pattern with the marking scheme.
Class 11 Chemistry Exam Pattern
In this Section, we have mentioned the Class 11 Chemistry Exam Pattern. Students can check the Class 11 Chemistry Exam Pattern for the academic year 2024-25.
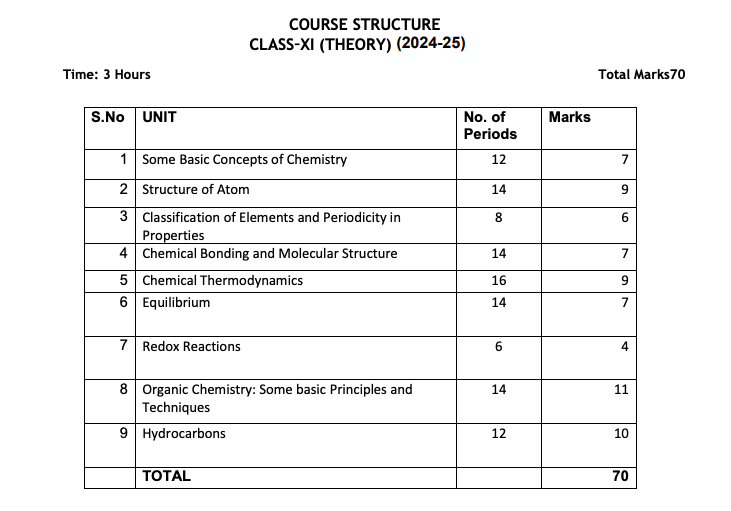
NOTE:- To learn more About the Class 12 New Chemistry Syllabus Session 2024-25
Class 11 Study Materials
We have tried to bring all the valuable links to CBSE NCERT Study Materials like Syllabus, Worksheet, Sample Paper, NCERT Solutions, Marking Scheme, Important Books, Holiday Homework, Previous Year Question Papers and Study Materials. You can visit all these important links by clicking the links/Icons.
